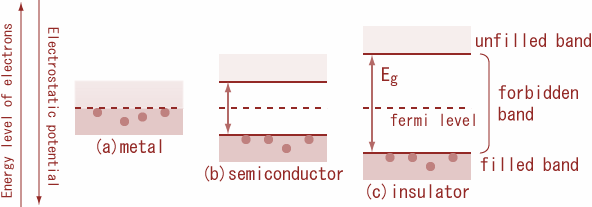
Carbon, silicon and germanium have four valence electrons each. These are characterised by valence and conduction bands separated by energy band gap respectively equal to ${\\left( {{E_g}} \\right)_C},{\\text{ }}{\\left( {{E_g}} \\right)_{Si}}{\\text ...

Exciton-driven change of phonon modes causes strong temperature dependent bandgap shift in nanoclusters | Nature Communications








![Solved [16 points] Silicon is a semiconductor with a band | Chegg.com Solved [16 points] Silicon is a semiconductor with a band | Chegg.com](https://media.cheggcdn.com/media/a84/a844b695-a87f-4466-9505-823ff317aefc/phpcfV989)
