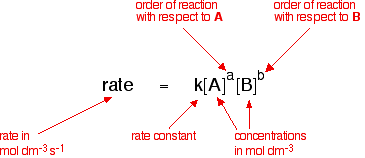
16.1 Rate expression Distinguish between the terms rate constant, overall order of reaction and order of reaction with respect to a particular reactant. - ppt download

How to Determine the Order of Reaction by Comparing Initial Rates of Reactions | Chemistry | Study.com

Determine the rate law for the overall reaction (where the overall rate constant is represented as k) - Home Work Help - Learn CBSE Forum
![I know the overall rate law is r=k[NO]^2[H2]^1, and that the RDS has the rate law in it with its coefficients, but there are 1) no speeds and 2) there are 2 I know the overall rate law is r=k[NO]^2[H2]^1, and that the RDS has the rate law in it with its coefficients, but there are 1) no speeds and 2) there are 2](https://preview.redd.it/i-know-the-overall-rate-law-is-r-k-no-2-h2-1-and-that-the-v0-we51rd60enda1.png?auto=webp&s=4ee8efdae71dba86eac26ee1ffe459617a39f65c)
I know the overall rate law is r=k[NO]^2[H2]^1, and that the RDS has the rate law in it with its coefficients, but there are 1) no speeds and 2) there are 2

Identifying the Rate Law for a Reaction from a Mechanism Where the First Step is Rate Limiting | Chemistry | Study.com

Determine the rate law for the overall reaction (where the overall rate constant is represented as k)? | Socratic




![16.1/R2.2.6 Rate constant, overall order of reaction, order of reaction [HL IB Chemistry] - YouTube 16.1/R2.2.6 Rate constant, overall order of reaction, order of reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/9sMFJMuZzmg/maxresdefault.jpg)