Explosives . H2O,H,S0, Fig. 28. Degree of Nitration of Nibro-cotton a a Function of tIh- Molecularmpoaition of Mixed Acid and the Vapour Tension f tin- Nitric Acid collodion cottons completely or almost
Materials | Free Full-Text | Phase Equilibria of the In–Pd–Sn System at 500 °C and 800 °C: Experimental Study and CALPHAD Modeling
Phase diagram and structural evolution of tin/indium (Sn/In) nanosolder particles: from a non-equilibrium state to an equilibrium state - Nanoscale (RSC Publishing) DOI:10.1039/C7NR01402C

Partial Palladium Oxidation over Various Oxide Supports for a Higher Reactivity of PdO with Respect to CH4 | The Journal of Physical Chemistry C

Thin Multilayer Palladium Coatings for Semiconductor Packaging Applications Part I: Solderability | Products Finishing
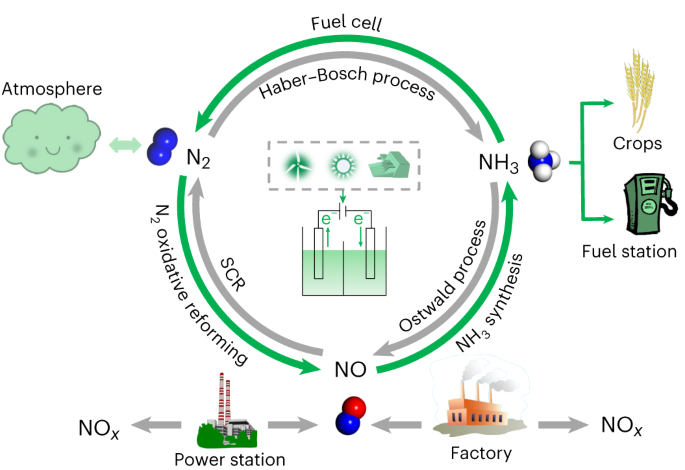
Electrochemical synthesis of ammonia from nitric oxide using a copper–tin alloy catalyst | Nature Energy

Alkaline Formate Oxidation with Colloidal Palladium–Tin Alloy Nanocrystals | ACS Applied Energy Materials

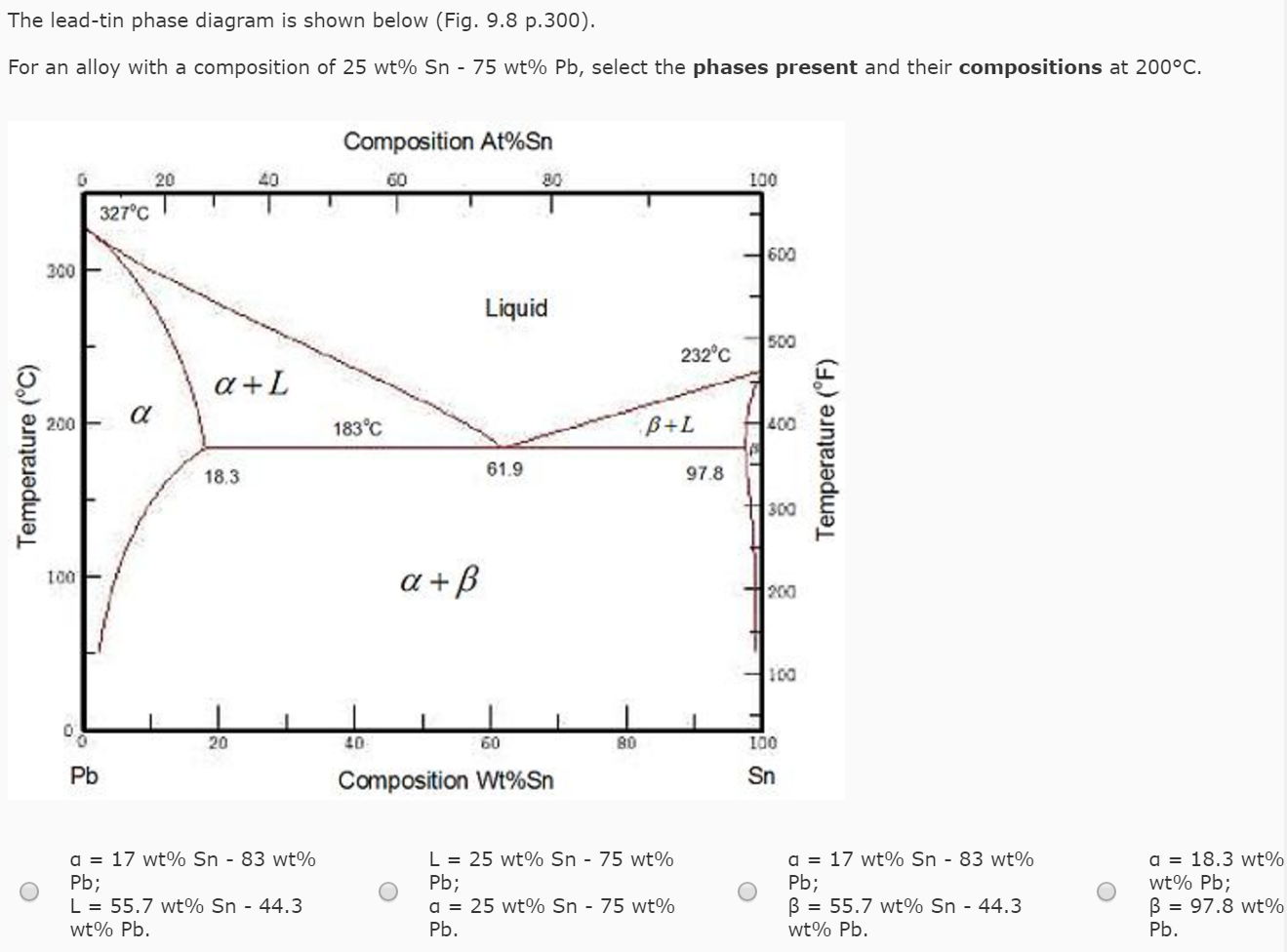
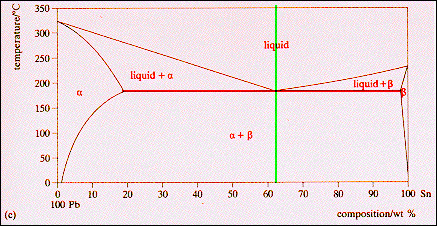
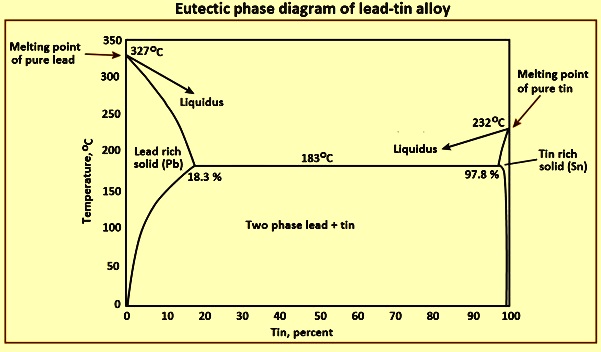
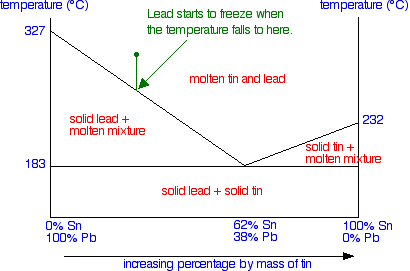
A lead-tin alloy of composition 90 wt% Sn - 10 wt% Pb is slowly heated from a temperature of 100C. At what temperature does the first liquid phase form? What is the

![1: Binary Pd-Sn phase diagram adapted from H. Okamoto [130]. | Download Scientific Diagram 1: Binary Pd-Sn phase diagram adapted from H. Okamoto [130]. | Download Scientific Diagram](https://www.researchgate.net/publication/321875056/figure/fig54/AS:631629873680428@1527603705926/Binary-Pd-Sn-phase-diagram-adapted-from-H-Okamoto-130.png)
![Phase diagram of the palladium-platinum binary metal system [1] | Download Scientific Diagram Phase diagram of the palladium-platinum binary metal system [1] | Download Scientific Diagram](https://www.researchgate.net/publication/348604433/figure/fig1/AS:1022471264272384@1620787554327/Phase-diagram-of-the-palladium-platinum-binary-metal-system-1.png)