Package leaflet: Information for the patient Pramipexol Teva Pharma 0.26 mg prolonged-release tablets Pramipexol Teva Pharma 0.5
Pramipexol Teva Pharma Läs noga igenom denna bipacksedel innan du börjar använda detta läkemedel. Den innehåller informatio
Písomná informácia pre používateľa Pramipexol Teva Pharma 0,26 mg Pramipexol Teva Pharma 0,52 mg Pramipexol Teva Pharma 1,

Pramipexole for the treatment of depressive symptoms in patients with Parkinson's disease: a randomised, double-blind, placebo-controlled trial - The Lancet Neurology
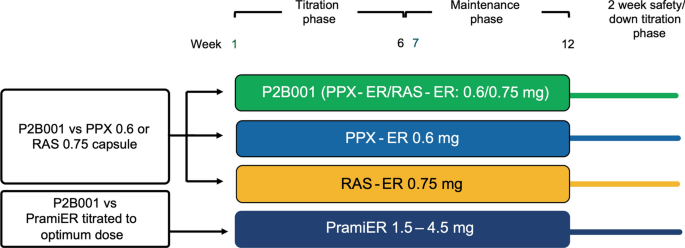
P2B001 (Extended Release Pramipexole and Rasagiline): A New Treatment Option in Development for Parkinson's Disease | Advances in Therapy

Teva Pharmaceutical Industries Limited (TEVA) - Pharmaceuticals & Healthcare - Deals and Alliances Profile | PDF | Initial Public Offering | Pharmaceutical Industry